Clinical research is scientific research conducted with the participation of humans. The aim of clinical research is to confirm the safety and effectiveness of new drugs or treatment methods. Carrying out clinical research is the only reliable way to establish whether a new treatment method is more effective than the methods applied so far.
CLINICAL RESEARCH
Scientific research conducted with the participation of humans with the aim to invent or confirm medicines’ effectiveness and safety
Definition included in the Pharmaceutical Law of September 6, 2001 (Journal of Laws of 2001 No. 126 item 1381, as amended):
“clinical research is each study conducted with the participation of humans with the aim to invent or confirm the clinical, pharmacological, including pharmacodynamic, effects of one or several researched medicinal products, or to identify the adverse effects of one or larger number of researched medicinal products, or to track the absorption, distribution, metabolism and excretion of one or larger number of researched medicinal products, taking into account their safety and effectiveness;”
SPONSOR
Definition included in the Pharmaceutical Law of September 6, 2001 (Journal of Laws of 2001 No. 126 item 1381, as amended):
“natural person, legal person or organizational unit without legal personality responsible for undertaking, conducting and financing a clinical study, with its registered office in the territory of one of the European Union member states or the European Free Trade Agreement (EFTA) member states – parties to the agreement on the European Economic Area; if the sponsor does not have its registered office in the territory of one of the European Economic Area member states, it may only act through its legal representative having its registered office in this territory;”
RESEARCH CENTER
A healthcare facility, e.g. a hospital or a health center, where clinical research is conducted.
Rules of conducting clinical studies
Clinical studies are conducted in accordance with strictly defined rules whose aim is to ensure maximum safety for the patient (participant of the study) and to respect his or her rights. It is the doctor performing the role of the researcher who decides whether the patient fulfils the medical criteria to take part in the study.
Participation in a clinical study is voluntary, and it requires suitable preparation as well as analysis of one’s decision. The researcher who is recruiting patients for the study must thoroughly explain all the aspects of the study which are unclear for the participant and which raise their doubts. A patient who decides to take part in the clinical study signs the so-called informed consent for participation in the clinical study.
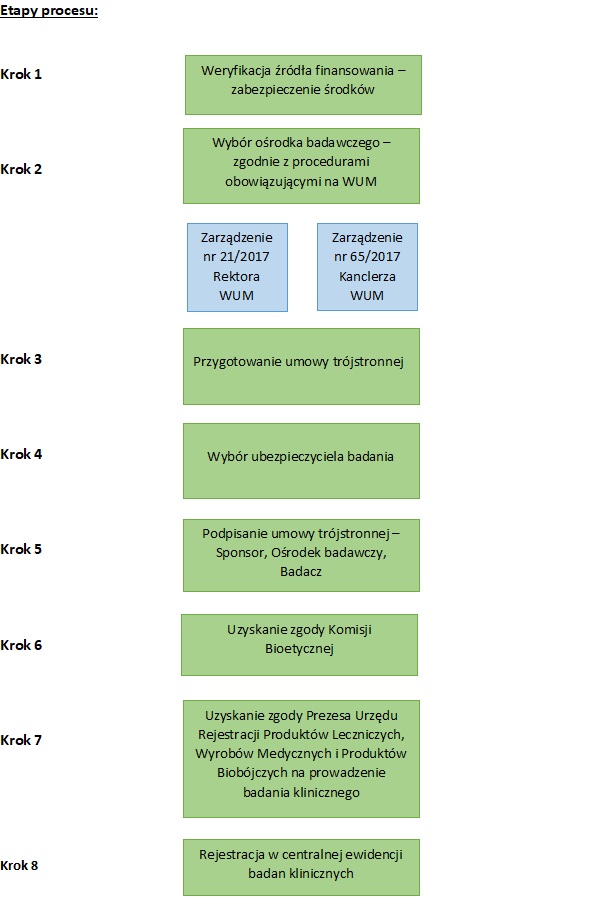
Clinical research at MUW
MUW may solely perform the role of the sponsor of clinical research. The source of financing clinical research are MUW’s own resources, the Ministry of Health’s subvention in research part, the projects of the National Center for Research and Development, National Science Centre, Ministry of Health, donations, pharmaceutical industry.
New opportunities for applying for non-commercial clinical research financing
Medical Research Agency
The Agency shall support the development of research in the field of medical sciences and health sciences as well as medical technology including, but not limited to non-commercial clinical studies of all phases of medicinal products and medical devices, as well as epidemiological research and interventions, analyses and modelling of large public databases in healthcare and research regarding management, development and optimization of the healthcare system. The key task of the Agency shall be analytical activity within the area of evaluating the impact of the management decisions taken on the costs of healthcare system functioning.
Control over the clinical study process
In Poland, the supervision over clinical studies and admission to trading of new medicines is performed by the President of the Office for Registration of Medicinal Products, Medical Devices and Biocidal Products. Competent Bioethics Committees issue their opinions on the ethics of the clinical study with particular attention paid to the rights of the study participant.
By clicking on the addresses listed below, you will be directed to the Internet websites containing official bases of clinical studies in different implementation phases, including also the studies conducted in Poland.
If needed, check the user manuals for the bases, which can be found next to the links.
- apps.who.int – World Health Organization (WHO), guidebook for download.
- www.clinicaltrials.gov - U.S. National Library of Medicine, guidebook for download. MUW Małgorzata Zawistowska - Samulczyk; malgorzata.zawistowska@wum.edu.pl tel. 22 572 04 66;
- www.clinicaltrialsregister.eu – European Medicines Agency (EMA), guidebook for download (in English).
REGISTERS OF CLINICAL STUDIES
Databases on clinical studies in English:
- Worldwide - https://clinicaltrials.gov
- European (EMA) - https://www.clinicaltrialsregister.eu/ctr-search/search
- WHO - www.who.int/ictrp/en (meta-register, i.e. it does not register studies on its own, but it collects them and displays them from several worldwide registers; except for the American and European ones, also from large national registers, including, but not limited to the Australian, Japanese, Chinese, Indian ones).
- Great Britain - https://ukctg.nihr.ac.uk/trials?query=%257B%2522query%2522%2B:%2B%2522*%2522%257D
- European Union post-authorization studies regarding safety (PASS studies) and pharmacoepidemiological studies - http://www.encepp.eu/encepp/studiesDatabase.jsp
- Poland Clinical Trials - https://www.centerwatch.com/clinical-trials/listings/location/international/Poland/
Databases on clinical studies in Polish:
- INFARMA - http://www.badaniaklinicznewpolsce.pl/baza-badan-klinicznych/
- „Badania Kliniczne w Polsce” (“Clinical Studies in Poland”) - www.bkwp.pl
- Immuno-Oncology Register - www.immuno-onkologia.pl
- Register of oncological studies - https://pto.med.pl/badania_kliniczne
- Polish Coalition of Oncological Patients - http://www.pkopo.pl/badania_kliniczne
- eBadania Kliniczne - https://ebadaniakliniczne.pl/
- Register of the Department for Early Phase Studies by the National Research Institute of Oncology - http://www.obwf.coi.pl
